Recall of blood pressure medications expanded for 5th time due to possible cancer risk

FDA expands blood pressure medication recall
The medication is used to treat hypertension, hypertensive patients with left ventricular hypertrophy and nephropathy in Type 2 diabetic patients.
WASHINGTON - Federal health officials said a recall for medication used to treat high blood pressure has been expanded for a fifth time after trace amounts of a possible cancer-causing substance were detected.
Torrent Pharmaceuticals Limited announced its expanded recall Thursday for losartan potassium tablets USP and losartan potassium/hydrochlorothiazide tablets, USP, due to the detection of trace amounts of an “unexpected impurity” while testing finished product batches, according to the U.S. Food and Drug Administration.
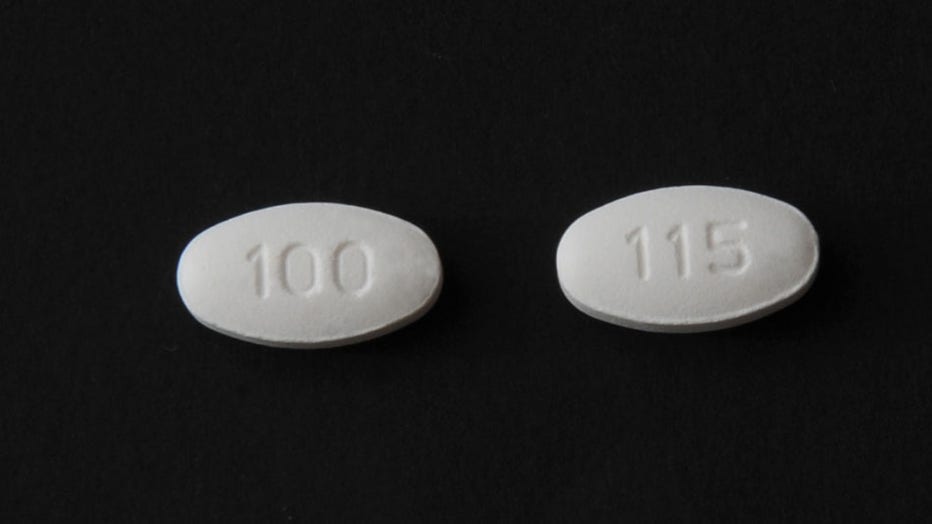
A provided image shows high blood pressure medication under the expanded recall, announced by the FDA. (Photo credit: Provided/FDA)
The substance, called N-Methylnitrosobutyric acid or NMBA, is a “probable human carcinogen,” according to the FDA.
The medication is used to treat hypertension, hypertensive patients with left ventricular hypertrophy and nephropathy in Type 2 diabetic patients, the FDA said.
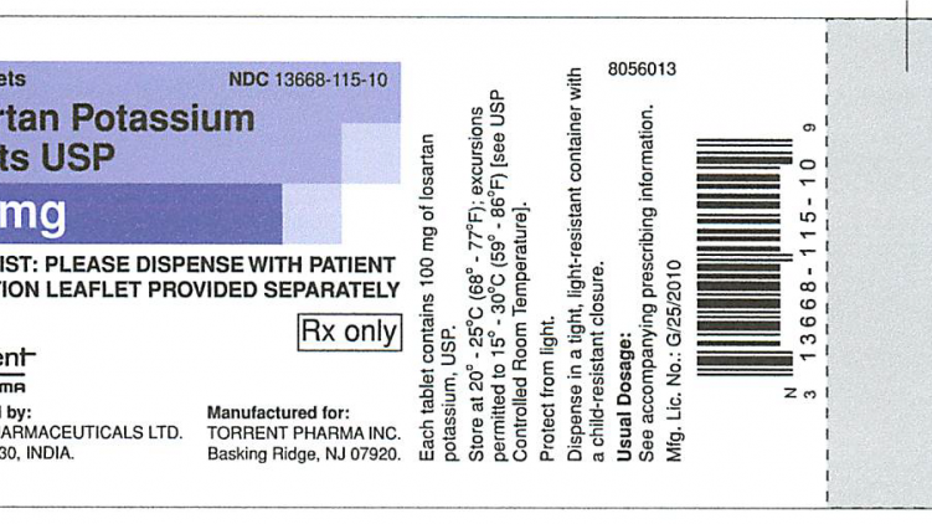
The medication under the expanded recall can be identified by checking the product name, manufacturer details and batch or lot number on the bottle, the FDA said. (Photo credit: Provided/FDA)
Federal health officials said patients should continue taking their medication as the risk of harm may be higher if the treatment is stopped immediately without an alternative. Patients should also contact their doctor about an alternative treatment.
RELATED: Generic Zantac makers halt worldwide distribution after FDA finds possible cancer chemical in drugs
The medication under the expanded recall can be identified by checking the product name, manufacturer details and batch or lot number on the bottle, the FDA said. A full product list is available online.
Drugmakers have issued dozens of recalls since July linked to low levels of the probable cancer-causing chemical.
The affected medications are low-cost versions of lifesaving heart-regulating drugs, including valsartan, losartan and irbesartan. They are designed to allow blood to flow more easily and are sold as single-ingredient pills and tablets and in combination with other drugs.
This story was reported from Cincinnati. The Associated Press contributed.

